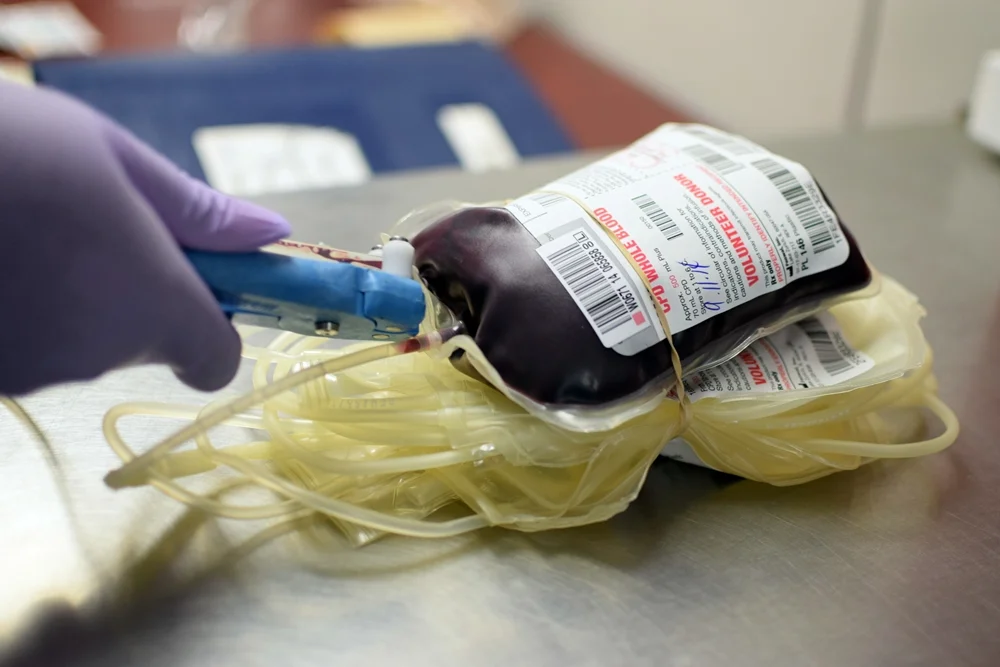
Whole Blood: Demand is on the Rise
/Blood centers across the country are seeing a renewed interest in whole blood transfusions. The main fuel to this fire appears to be the recent change from AABB and the forthcoming 31st edition of Standards for Blood Banks and Transfusion Services which allows for the use of low titer group O whole blood in emergent situations for recipients of unknown blood type (AABB News, 2017).
Read More